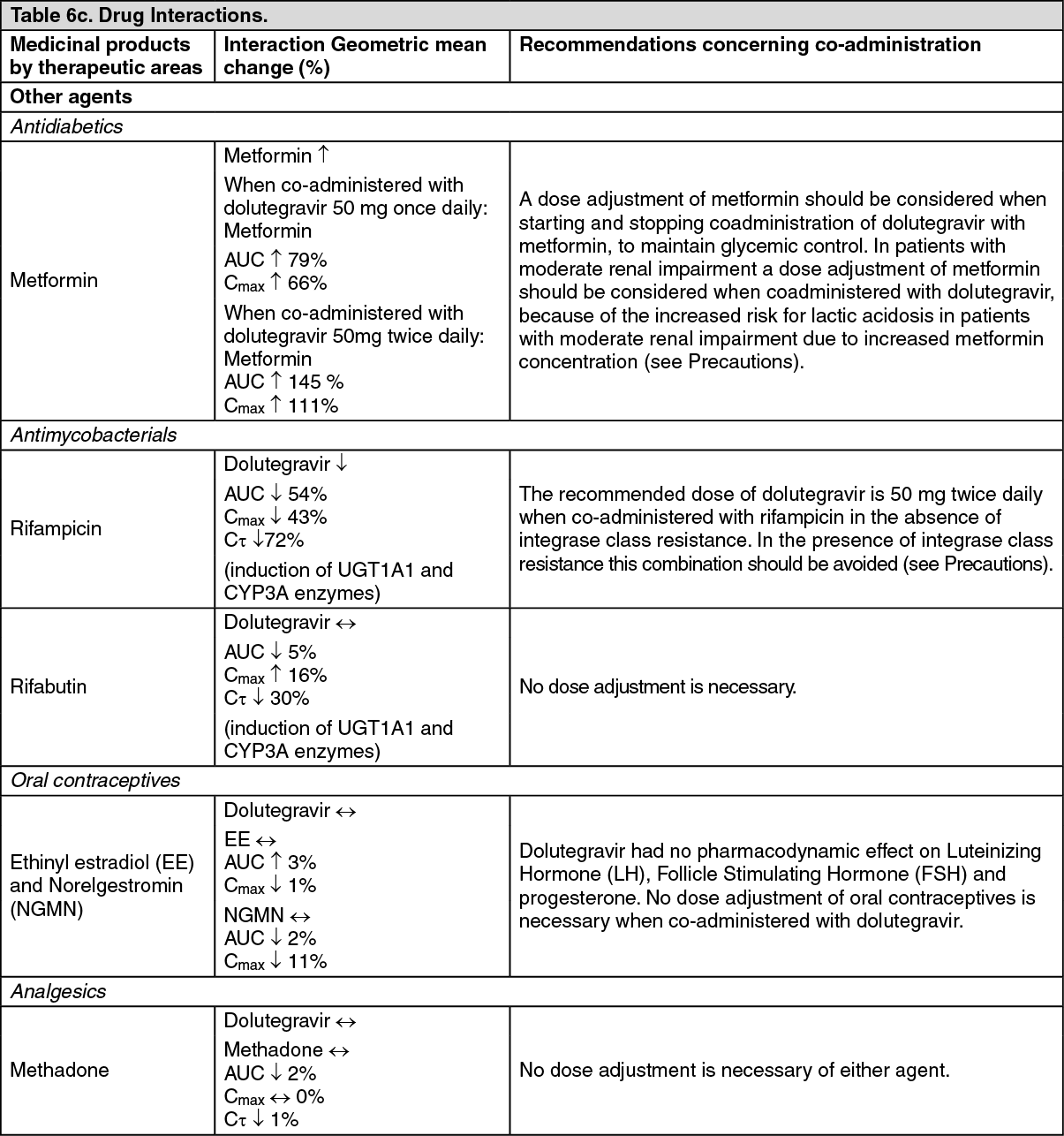
AUSTRALIAN PRODUCT INFORMATION TIVICAY (dolutegravir) film-coated tablets 1 NAME OF THE MEDICINE 2 QUALITATIVE AND QUANTITATIVE

Dosing Considerations | See Boxed Warning for TRIUMEQ & TRIUMEQ PD (abacavir, dolutegravir, lamivudine)

PDF) Simplified dolutegravir dosing for children with HIV weighing 20 kg or more: pharmacokinetic and safety substudies of the multicentre, randomised ODYSSEY trial

Figure 2 from Once daily dolutegravir (S/GSK1349572) in combination therapy in antiretroviral-naive adults with HIV: planned interim 48 week results from SPRING-1, a dose-ranging, randomised, phase 2b trial. | Semantic Scholar

SINGLE AND MULTIPLE DOSE DOLUTEGRAVIR PHARMACOKINETICS IN THE GENITAL TRACT AND COLORECTUM OF HIV NEGATIVE MEN AND WOMEN

Dolutegravir dosing for children with HIV weighing less than 20 kg: pharmacokinetic and safety substudies nested in the open-label, multicentre, randomised, non-inferiority ODYSSEY trial - The Lancet HIV

Dolutegravir-based and low-dose efavirenz-based regimen for the initial treatment of HIV-1 infection (NAMSAL): week 96 results from a two-group, multicentre, randomised, open label, phase 3 non-inferiority trial in Cameroon - The Lancet

Table 5 from Dolutegravir (DTG) Based Fixed Dose Combination (FDC) of Tenofovir/Lamivudine/Dolutegravir (TLD) and Viral Load Suppression in Children in Port Harcourt, Nigeria | Semantic Scholar

Simplified dolutegravir dosing for children with HIV weighing 20 kg or more: pharmacokinetic and safety substudies of the multicentre, randomised ODYSSEY trial - ScienceDirect

KIEV, UKRAINE - September 25, 2019: Dolutegravir / Lamivudine is the First Two-component Drug in a Fixed Dosage, Which is a Comple Editorial Image - Image of complex, editorial: 158426830

A single dose of ultra-LA dolutegravir protects against multiple high... | Download Scientific Diagram

Simplified dolutegravir dosing for children with HIV weighing 20 kg or more: pharmacokinetic and safety substudies of the multicentre, randomised ODYSSEY trial - The Lancet HIV
Clinical effectiveness of dolutegravir in the treatment of HIV/AIDS - Document - Gale Academic OneFile
AUSTRALIAN PRODUCT INFORMATION DOVATO (dolutegravir/lamivudine fixed-dose combination) film-coated tablets 1 NAME OF THE MEDICIN

New approaches for paediatric dosing: abacavir in newborns, doubling dolutegravir with rifampicin | aidsmap

Dolutegravir twice-daily dosing in children with HIV-associated tuberculosis: a pharmacokinetic and safety study within the open-label, multicentre, randomised, non-inferiority ODYSSEY trial - The Lancet HIV

Metabolism, Excretion, and Mass Balance of the HIV-1 Integrase Inhibitor Dolutegravir in Humans | Antimicrobial Agents and Chemotherapy