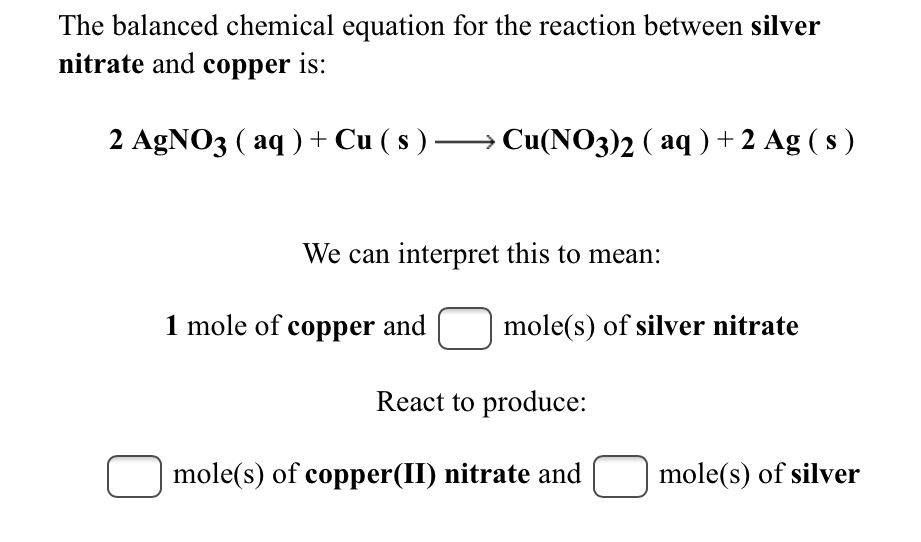
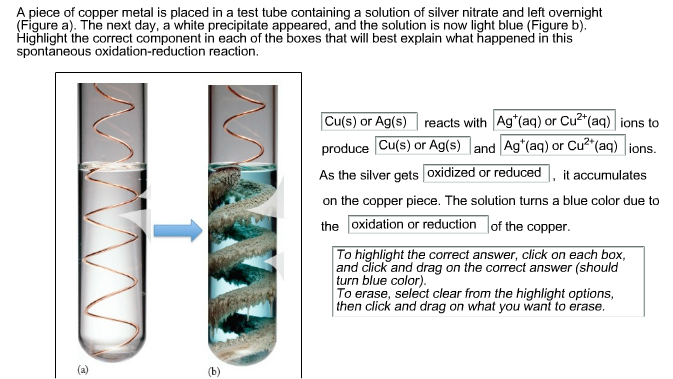
When copper is dipped in the solution of silver nitrate, the solution turns blue. Give the reason along with chemical equation?

SOLVED: Table 6.3 This photo shows the results of putting copper metal into a solution of silver nitrate. The progress of the reaction is shown: Copper initial Silver nitrate initial After mixing





/copper-wire-immersed-in-silver-nitrate-causing-blue-colour-81991997-582f14595f9b58d5b1a9b484.jpg)